By Kevin Hein PA-C
When it comes to the ever-expanding world of pharmacology (the study of drugs), the vast number of brand name and generic drugs can be extremely confusing for healthcare providers, let alone their patients. Many patients have had the experience of being prescribed one medication at their appointment, only to discover a different name on their prescription bottle when they get home. As healthcare providers, it is important that we try to make patients comfortable with the drugs we prescribe. Understanding the difference between brand name and generic drugs is an important part of that discussion.
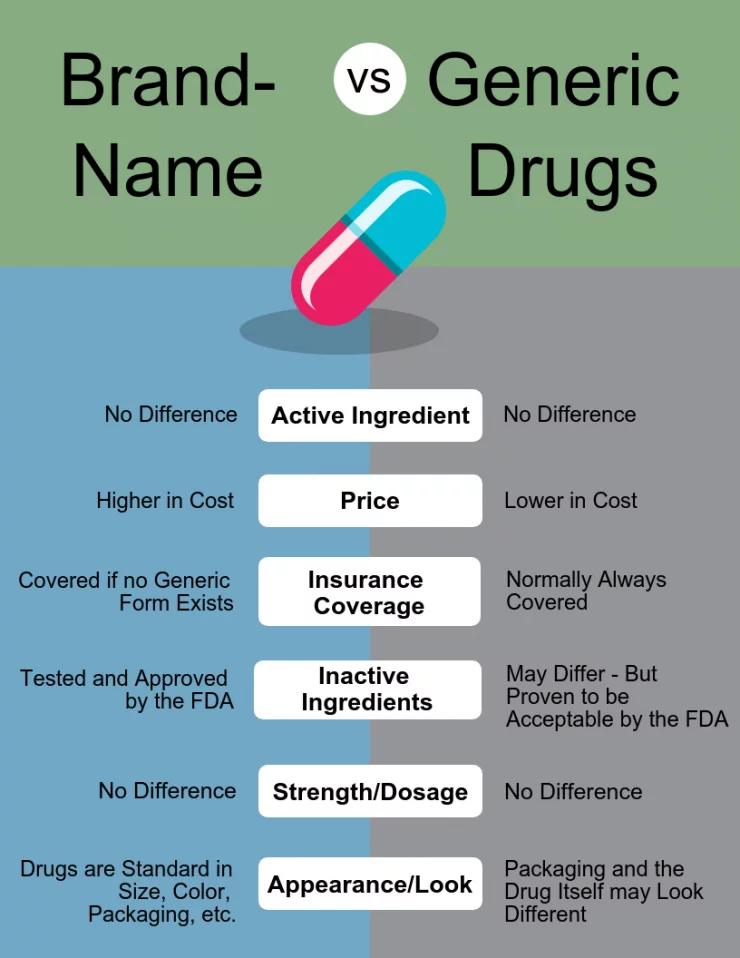
So what is the difference between brand name and generic drugs? In short, not a lot.
• Contain the same active ingredient
• Same level of safety, efficacy, and adverse effects.
• Differ in their appearance (size, shape, color)
• Inactive ingredients are usually different.
• Cost
Your healthcare provider may refer to a drug by its brand name, but it is the generic version that we most often prescribe. In fact, it is state-law that if we prescribe a medication and there is a generic version available, the pharmacy must fill the generic version of the drug unless we say otherwise. This is due to the fact that the generic version is almost always lower in cost than the brand name drug. Why is that?
Let’s start by looking at the drug development process.
• Discovery or development of new drug through scientific research
• Obtaining US patent
• Clinical trials
• FDA approval, “Brand-naming”, and marketing to the public
When a pharmaceutical company discovers or designs a potential new drug, it incurs significant cost in doing research, development, and clinical trials. As part of the process, the pharmaceutical company obtains a patent for the drug and once it is approved by the Food and Drug Administration (FDA), they market the drug to the public. This is where the drug gets its “Brand name.” The marketing branch of a pharmaceutical company applies a easy-to-pronounce, memorable name to their drug to help sell it. Names like Advair and Abilify roll off the tongue much better than their actual drug names - fluticasone-salmeterol and aripiprazole.
As with many other commercial products, US patents for brand name drugs have term limits, generally about 20 years. When the brand name drug is the only version available on the market, the price is often quite high. This is because the pharmaceutical company needs to cover the cost of the research, development, and clinical trials of the drug, and then turn a profit. Brand name drugs are often marketed as being superior to alternative drugs to treat disease and can be attractive to both patients and healthcare providers.
Once the patent on the brand name drug expires, other pharmaceutical companies are allowed to produce their own version of the drug. These versions are known as the “Generic” versions. Just like the brand name drug, the generic drug needs to be approved by the FDA, but the companies that produce generic versions of a drug are able to sell them for much less than the brand name drug, as they did not incur any costs for research or clinical trials. There is no limit to the number of generic versions of a drug that can be produced, so there are often several pharmaceutical companies that will produce generic versions of a brand name drug. This creates competition in the market and drives prices lower.
If your doctor prescribes you a medication that is only available in the brand name form, you will likely find it to be very expensive. Insurance companies are often hesitant to include brand name drugs in their formularies due to cost. If your doctor feels that it is absolutely medically necessary for you to be taking the brand name drug, they can appeal to your health insurance company for the brand name drug to be covered by insurance. This is called Prior Authorization. If coverage of the drug is still denied, the pharmaceutical company will often have coupons available for their brand name drugs for patients with commercial insurance. This can help offset the cost of the brand name drug considerably.
It is important to remember the pharmaceutical companies are in the business to make money, but also be aware that it is in their best interest to continue to discover and design drugs that improve the quality of our healthcare. Drugs that were well designed 50 years ago are still used regularly today. I always encourage my patients to ask questions about their medications and to know them well. Make sure that you and your healthcare provider practice informed-decision making when it comes to your brand name and generic drugs.